IMPORTANT SAFETY INFORMATION
UDENYCA (pegfilgrastim-cbqv) is contraindicated in patients with a history of serious allergic reactions to pegfilgrastim products or filgrastim products. Reactions have included anaphylaxis.
Splenic rupture: Splenic rupture, including fatal cases, can occur following administration of pegfilgrastim products. Evaluate patients who report left upper abdominal or shoulder pain for an enlarged spleen or splenic rupture.
Acute respiratory distress syndrome (ARDS): Evaluate patients who develop fever, lung infiltrates, or respiratory distress. Discontinue UDENYCA in patients
with ARDS.
Serious allergic reactions, including anaphylaxis: The majority of reported events occurred upon initial exposure. Allergic reactions, including anaphylaxis, can recur within days after the discontinuation of initial anti-allergic treatment. Permanently discontinue UDENYCA in patients with serious allergic reactions.
Allergies to acrylics (UDENYCA ONBODY® only): The on-body injector (OBI) for UDENYCA uses acrylic adhesive. For patients who have reactions to acrylic adhesives, use of this product may result in a significant reaction.
Sickle cell disorders: Severe and sometimes fatal crises have occurred in patients with sickle cell disorders. Discontinue UDENYCA if sickle cell crisis occurs.
Glomerulonephritis: The diagnoses were based upon azotemia, hematuria (microscopic and macroscopic), proteinuria, and renal biopsy. Generally, events resolved after dose reduction or discontinuation. Evaluate and consider dose-reduction or interruption of UDENYCA if causality is likely.
Leukocytosis: White blood cell (WBC) counts of 100 x 109/L or greater have been observed in patients receiving pegfilgrastim products. Monitoring of complete blood count during UDENYCA therapy is recommended.
Thrombocytopenia: Thrombocytopenia has been reported in patients receiving pegfilgrastim products. Monitor platelet counts.
Capillary leak syndrome: Has been reported after granulocyte colony stimulating factor (G-CSF) administration, including pegfilgrastim products, and is characterized by hypotension, hypoalbuminemia, edema, and hemoconcentration. Episodes vary in frequency and severity and may be life-threatening if treatment is delayed. If symptoms develop, closely monitor and give standard symptomatic treatment, which may include a need for intensive care.
Potential for tumor growth stimulatory effects on malignant cells: The G-CSF receptor through which pegfilgrastim products and filgrastim products act has been found on tumor cell lines. The possibility that pegfilgrastim products act as a growth factor for any tumor type, including myeloid malignancies and myelodysplasia, diseases for which pegfilgrastim products are not approved, cannot be excluded.
Myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML) in patients with breast or lung cancer: MDS and AML have been associated with the use of pegfilgrastim products in conjunction with chemotherapy and/or radiotherapy in patients with breast or lung cancer. Monitor patients for signs and symptoms of MDS/AML in these settings.
Potential device failures (UDENYCA ONBODY® only): Missed or partial doses have been reported in patients receiving UDENYCA via the OBI due to the device not performing as intended. In the event of a missed or partial dose, patients may be at increased risk of events such as neutropenia, febrile neutropenia and/or infection than if the dose had been correctly delivered. Instruct patients using the OBI to notify their healthcare professional immediately to determine the need for a replacement dose of UDENYCA if they suspect that the device may not have performed as intended.
Aortitis: Has been reported in patients receiving pegfilgrastim products, occurring as early as the first week after start of therapy. Manifestations may include generalized signs and symptoms such as fever, abdominal pain, malaise, back pain, and increased inflammatory markers (eg, c-reactive protein and WBC count). Consider aortitis when signs and symptoms develop without known etiology. Discontinue UDENYCA if aortitis is suspected.
Nuclear imaging: Increased hematopoietic activity of the bone marrow in response to growth factor therapy has been associated with transient positive bone imaging changes. Consider when interpreting bone imaging results.
The most common adverse reactions (≥5% difference in incidence compared to placebo) are bone pain and pain in extremity.
INDICATION
UDENYCA is a leukocyte growth factor indicated to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs associated with a clinically significant incidence of febrile neutropenia.
Limitation of Use
UDENYCA is not indicated for the mobilization of peripheral blood progenitor cells for hematopoietic stem cell transplantation.
To report SUSPECTED ADVERSE REACTIONS, contact Accord BioPharma Inc at 1-866-941-7875 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
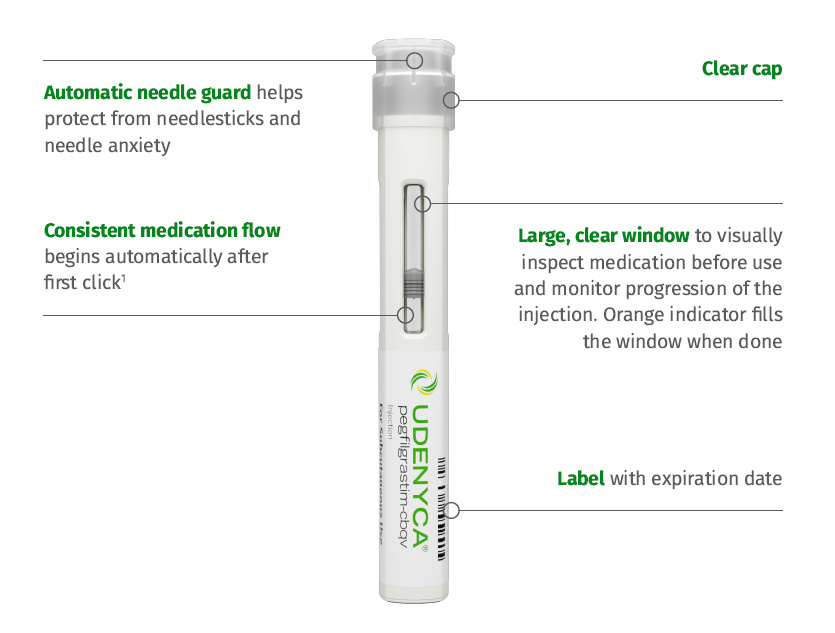
UDENYCA® is supplied as:
- Prefilled syringe: 6 mg/0.6 mL in a single-dose prefilled syringe for manual use only
- Autoinjector: 6 mg/0.6 mL in a single-dose prefilled autoinjector
- UDENYCA ONBODY®: 6 mg/0.6 mL in a single-dose prefilled syringe co-packaged with the on-body injector for UDENYCA®
For more information, please see the full Prescribing Information.
References: 1. Klastersky J, de Naurois J, Rolston K, et al. Management of febrile neutropenia: ESMO clinical practice guidelines. Ann Oncol. 2016;27:111-V118. https://www.annalsofoncology.org/article/S0923-7534(19)31643-6/fulltext. 2. Weycker D, Li X, Edelsberg, et al. Risk and consequences of chemotherapy-induced febrile neutropenia in patients with metastatic solid tumors. Oncol Pract. 2015;11:47-54. https://pubmed.ncbi.nlm.nih.gov/25491042/. 3. Culakova E, Thota R, Poniewierski MS, et al. Patterns of chemotherapy-associated toxicity and supportive care in US oncology practice: a nationwide prospective cohort study. Cancer Med. 2014;3:434-444. https://doi.org/10.1002/cam4.200. 4. Boccia R, Glaspy J,Crawford J, et al. Chemotherapy-induced neutropenia and febrile neutropenia in the US: a beast of burden that needs to be tamed? The Oncologist. 2022;27(8):625-636. https://pubmed.ncbi.nlm.nih.gov/35552754/. 5. Pathak R, Giri S, Aryal MR, et al. Mortality, length of stay, and healthcare costs of febrile neutropenia-related hospitalizations among patients with breast cancer in the United States. Support Care Cancer. 2015;23:615-617. https://pubmed.ncbi.nlm.nih.gov/25556610/. 6. Tai E, Guy GP, Dunbar A, et al. Cost of cancer-related neutropenia or fever hospitalizations, United States, 2012. Oncol Pract. 2017;13:552-561. https://pubmed.ncbi.nlm.nih.gov/28437150/#:~:text=For%20adults%2C%20the%20mean%20length,cost%20of%20%2426%2C000%20per%20stay. 7. Li S, Liu J, Bowers C, et al. Febrile neutropenia-related care and associated costs in elderly patients with breast cancer, lung cancer, or non-Hodgkin lymphoma. Support Care Cancer. 2020;28:113-122. https://doi.org/10.1007/s00520-019-04795-0. 8. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Hematopoietic Growth Factors V.3.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed February 14, 2024. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 9. Kuderer, M. Impact of primary prophylaxis with granulocyte colony-stimulating factor on febrile neutropenia. Clin Oncol. 2007;25(21):1-10. 10. UDENYCA (pegfilgrastim-cbqv) Prescribing Information. Accord BioPharma, Inc. 11. Neulasta® (pegfilgrastim) Prescribing Information. Amgen Inc.