UDENYCA is a treatment for febrile neutropenia
Chemotherapy can lower a patient’s white blood cell count, which can lead to febrile neutropenia, a common and
potentially life-threatening complication.1-3
For patients with cancer who visit the emergency
department for febrile neutropenia,
end in hospitalization4
Febrile neutropenia can cause treatment
delays and dose reductions,
negative outcomes1

UDENYCA is highly similar
to Neulasta® (pegfilgrastim)
Average length of hospital stays due
to febrile neutropenia
- 5.7 to 9.6 days (for adults)5,6
- 6.2 to 6.8 days (for elderly patients)7
Mortality rates in adults with cancer hospitalized for neutropenia/febrile neutropenia
- 2.6% to 7.0% (solid tumors)4,5
- 7.4% (hematologic malignancies)4
Factors driving febrile neutropenia hospitalization include4
- Older age
- Presence of comorbidities
- Advanced-stage disease
- First-cycle chemotherapy with average relative dose intensity ≥853,4
Febrile neutropenia contributes to rising healthcare costs
$2.3 BILLION
Total cost of hospitalizations for neutropenia
among adult patients with cancer6
$20K–$40K
Mean cost of febrile neutropenia
hospitalization5,6
NCCN Clinical Practice Guidelines in Oncology
(NCCN Guidelines®)
recommendation for G-CSF use during chemotherapy8
G-CSF (eg, pegfilgrastim) primary prophylaxis may support optimal dose intensity and clinical outcomes in certain patients
undergoing myelosuppressive chemotherapy.8
When febrile neutropenia risk is >20%, G-CSF prophylaxis9*:

Reduced the risk of febrile neutropenia9
(RR: 0.54; 95% CI, 0.43-0.67)

Significantly improved the relative dose intensity (RDI) of chemotherapy with an average difference in
RDI of 8.4%9
(G-CSF–treated patients vs non–G-CSF–treated patients)
G-CSF was shown to increase RDI and is associated with survival benefit.9†
*Results from a systematic review of 17 randomized trials, which included 3493 patients with solid tumors and lymphoma. G-CSF included filgrastim and pegfilgrastim.
†Survival benefit not included in the UDENYCA Prescribing Information.
CI, confidence interval; G-CSF, granulocyte colony-stimulating factor; RR, relative risk.
How UDENYCA works
UDENYCA helps stimulate the growth of white blood cells, which may help a patient

Fight of infection

Prevent febrile neutropenia
UDENYCA confirmed highly similar to
Neulasta® (pegfilgrastim)10,11
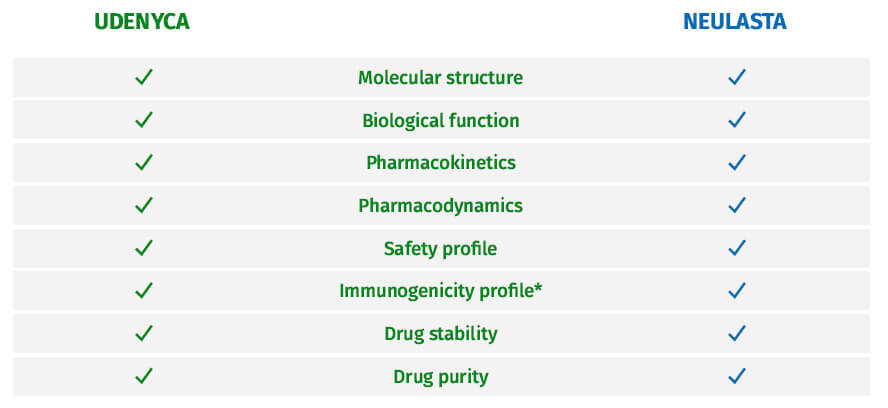
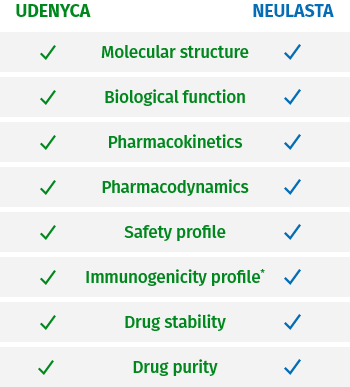
*The assays used to assess immunogenicity for UDENYCA were highly sensitive and demonstrated no treatment-emergent
neutralizing antibodies in more than 300 subjects.
Approval backed by data and FDA requirements12
Clinical studies with UDENYCA were conducted in healthy subjects, the most sensitive and informative population in which to assess biosimilarity.13
- A study in healthy subjects is likely to produce less pharmacokinetic/pharmacodynamic variability compared to a study in patients with potential confounding factors, such as underlying and/or concomitant disease and concomitant medications, such as myelosuppressive chemotherapy
- Healthy subjects are fully immunocompetent and can mount an immune antidrug antibody response
- Lack of confounding factors in healthy subjects allows for better evaluation of treatment-related adverse events that are specific to pegfilgrastim
Clinical studies in cancer patients to confirm biosimilarity of UDENYCA to Neulasta® were not required by the FDA.